Using complex state-of-the-art mouse models of cancer to improve the understanding and treatment of human cancer
Aims
Cancer remains one of the most prevalent health conditions for our population and 1 in 4 people in the UK currently die of the disease. Cancer is a complex and dynamic process which involves the transition of a normal cell into one which grows limitlessly through a series of changes in DNA and the surrounding tissues within which the tumour must survive and grow. These complex interactions are a vital component of how cancer develops; they influence whether cancer can spread through the body and whether it responds to treatment.
Research in cancer needs to be done using experimental tools that fully rebuild and maintain these tumour-tissue interactions. Mouse models of spontaneous cancer development are the best experimental systems, but for some cancers these models have fallen short of replicating the complexity of human disease. There is a need to develop the next generation of mouse models to better understand the biology underpinning the major cancer types, generate novel targets and rigorously apply these to a therapeutic pipeline with better prediction of patient response.
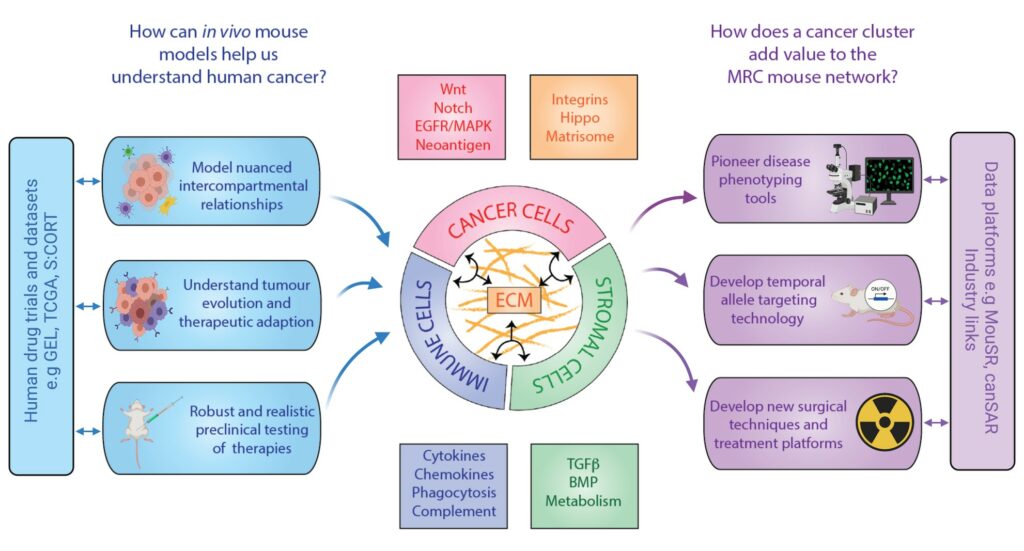
This cluster will exploit technologies that will allow us to characterise how accurately mouse models reflect specific subsets of human cancers, build better mouse models that mirror the full spectrum of human tumour evolution and that significantly improve our ability to predict therapeutic outcomes. We will focus on colorectal, lung, pancreatic and childhood cancers in key exemplar studies. We will create a compendium of models and data for the UK community, opening out availability of resources, technologies and expertise. With access to large human datasets we can refine the mouse models to faithfully reflect human disease and accurately predict therapeutic outcomes.
We will develop platforms for integrating various types of data across our spectrum of models, increasing their value as predictive preclinical tools. Specialist expertise in surgery, radiotherapy, imaging and standard-of-care treatment will be centralised through the Mary Lyon Centre, which will act as a repository of models for wider distribution and will promote best practice and 3Rs nationally. We aim to expand upon our established links with industry to promote commercialisation of new therapeutic discoveries and to leverage further significant investment in disease modelling through collaborative engagement. We expect to translate new treatments and drug combinations validated in key cancer models into the clinic through our extensive clinical network.
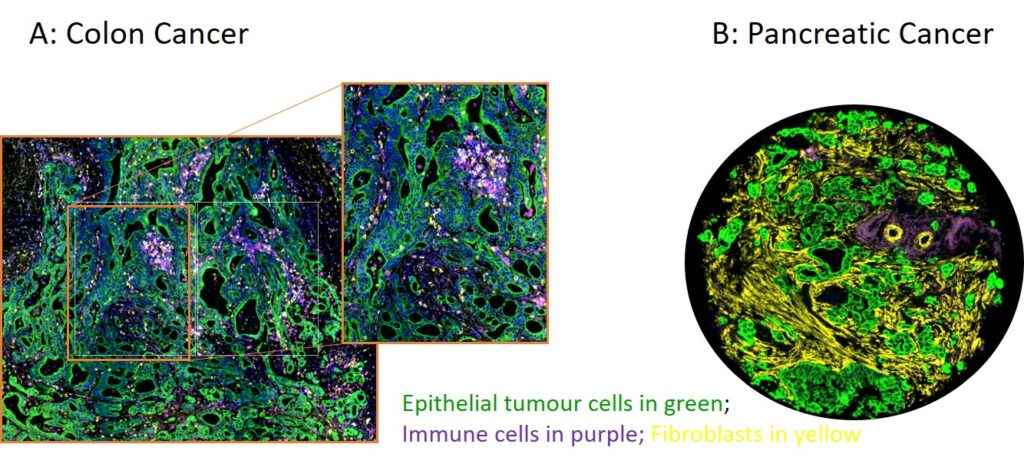
Spatial phenotyping of colon cancer (A) and pancreatic cancer (B) nicely illustrates how tumour cells are embedded in a dynamic microenvironment with cellular crosstalk between non-epithelial compartments such as fibroblasts and immune cells. Using molecular phenotyping techniques we can isolate these components to explore the signalling pathways key to tumour progression.
How we do it
Of all disease areas, cancer has one of the poorest records for converting preclinical leads into successful new therapies. Despite extensive investment and large consortia that have undertaken genomic profiling of cancers, there has been a disappointing paucity of new advances in cancer treatments. The emergence of deep molecular phenotyping datasets of human cancers has, however, allowed the recent development of new models of certain cancers that recapitulate the human disease with increasing accuracy in terms of both their genetics and their transcriptional signatures. These few exemplars have highlighted the need for the scientific community to disease-position mouse models of cancer that will allow us to better understand the biology underpinning the major cancer types, generate novel targets and rigorously apply these to a therapeutic pipeline with better prediction of patient response.
The Cancer Cluster will address this on a network scale, combining clinical, biological and computational expertise to undertake deep (molecular and spatial) phenotyping and disease-positioning, comparing across species and different cancers. In addition we will harness novel strategies (in harmony with NMGN expertise) to create sophisticated models that allow for temporal regulation of gene activation, to model new cancer drivers, and to target inhibition using protein degradation to more accurately align with future drug discovery efforts. Of key importance to our aims will be engagement with the UK network, achieved through the delivery of an easily accessible repository of fully phenotyped models, and the establishment of clinically relevant preclinical trials platforms based at the Mary Lyon Centre. Our cluster will work closely with academic, clinical and pharmaceutical partners to enable relevant preclinical trials, facilitating translation into new clinical trials throughout the course of this funding cycle.
Cluster Members
| Name | Organisation | Contact |
|---|---|---|
| Prof Karen Blyth | CRUK Scotland Institute / University of Glasgow | Karen.Blyth@glasgow.ac.uk |
| Prof Louis Chesler | Institute of Cancer Research | Louis.Chesler@icr.ac.uk |
| Dr Seth Coffelt | CRUK Scotland Institute / University of Glasgow | Seth.Coffelt@glasgow.ac.uk |
| Dr Philip Dunne | Queen’s University Belfast | p.dunne@qub.ac.uk |
| Dr Chiara Gorrini | Institute of Cancer Research | chiara.gorrini@icr.ac.uk |
| Prof Simon Leedham | University of Oxford | simon.leedham@well.ox.ac.uk |
| Prof Jennifer Morton | CRUK Scotland Institute / University of Glasgow | jennifer.morton@glasgow.ac.uk |
| Prof Daniel Murphy | CRUK Scotland Institute / University of Glasgow | daniel.murphy@glasgow.ac.uk |
| Prof Ian Tomlinson | University of Oxford | ian.tomlinson@oncology.ox.ac.uk |
Member Organisations

Cancer Research UK Scotland Institute

University of Glasgow

Institute of Cancer Research

Queen’s University Belfast


